Biosecure ID Investor Update | One Critical Year at a Time
Introduction
Over the last 12 months, Biosecure ID has crossed a vital execution threshold: from concept and early execution into real-world delivery, partnerships, and system-level innovation.
We strengthened our leadership and advisory bench, welcomed new colleagues, shipped products, closed meaningful commercial deals, and advanced a differentiated automation roadmap that addresses one of the most persistent challenges in applied genomics and forensic science: scalable, validated, end-to-end sample-to-answer workflows.
Yet the tension is real.
The problem we are solving is urgent, complex, and capital-intensive. Without the right infrastructure — a dedicated workshop, engineering capacity, validation environments, and manufacturing readiness — innovation risks stalling precisely at the moment when market pull is strongest.
This is the stage we are in today:
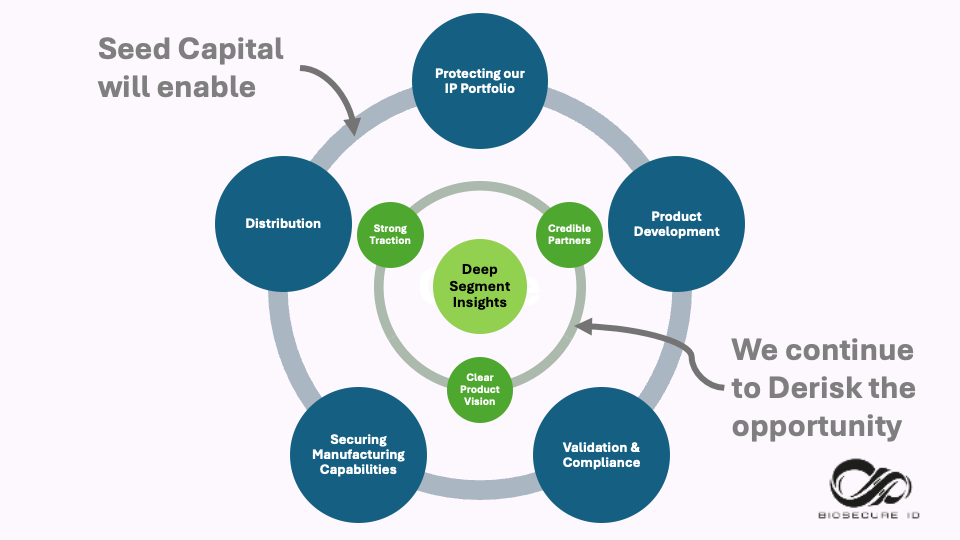
✔ strong traction: Shipped our products and delivered services to over 30 customers globally.
✔ credible partners in sample collection, Forensic DNA testing kits and instruments, Major liquid handling systems, and advanced AI-enabled software suppliers.
✔ clear product vision with its Vellixis Programme for Automated DNA Analysis of complex DNA samples obtained from victims and the scene of crime.
✖ constrained by capital at a critical inflection point
Task: What Needs to Change
Despite this momentum, the challenge ahead is clear: we must scale our automated solutions, complete prototyping and rigorous validation, and move toward mass production. Our goal is to launch our fully realised platform by March 2027, a timeline that hinges on funding to support product development, manufacturing readiness, and market entry.
This is where our story intersects with our audience — investors and partners — and their ability to help propel Biosecure ID into our next chapter.
Obstacles: What’s Standing in the Way
Transformative innovation in genomics and automation is complex. Obstacles include:
-
Engineering robust and scalable prototypes that meet regulatory and field standards.
-
Securing production capacity, supply chain resilience, and manufacturing partners.
-
Extending adoption across diverse sectors with varying technical and operational requirements.
-
The capital intensity of building automation and mass-production capabilities.
These obstacles are not abstract; they represent the very friction points that our technology, team, and partners are uniquely positioned to resolve.
Resolution: How We Change the Game
We are building solutions that are not incremental improvements — but fundamental shifts in how genomic evidence and forensic workflows operate. Customer deployments and active partner discussions are already shaping these requirements. Our roadmap over the next 12 months focuses on:
-
Fine-tuning and scaling our automated platforms through targeted workshops and systems engineering.
-
Completing prototypes and validation to ensure performance and reproducibility aligned with market needs.
-
Building the foundations for mass production capability.
-
Expanding strategic collaborations and commercial engagements that de-risk adoption while creating scalable revenue pathways.
By aligning technology development with customer needs and market realities, we are positioning Biosecure ID not just as a vendor but as a transformational partner to laboratories, forensic services, public health agencies, and enterprise genomics users.
Why We Are Uniquely Positioned
Biosecure ID is more than an idea. We are a team with deep segment expertise, a growing portfolio of products entering the market, validated partnerships with industry innovators, and a strategic investor narrative that aligns with long-term impact.
Our advantage lies not in a single component, but in owning the validation, automation, and deployment of the entire workflow — an execution problem incumbents struggle to solve. Our Team brings decades of experience building, validating and scaling applied genomics technologies in regulated environments.
For investors, the benefit is clear:
-
A compelling vision backed by tangible month-over-month advancements.
-
Real commercial traction and partnerships that demonstrate market pull.
-
A scalable technology roadmap with a clear product and production launch timeline — March 2027.
-
A business designed to change the market dynamics, not just compete within them.
Why is this the moment to engage
This round is designed with a clear techno-commercial intent:
-
To unlock the next phase of automation, validation, and production readiness
-
To convert momentum into defensible execution
-
To position the company for a credible, on-time launch in March 2027
For investors, the “Yes” is not about funding an idea — it is about enabling a system that is already taking shape, at the exact moment when speed, focus, and capital matter most. This round materially de-risks technical validation, manufacturing readiness, and commercial adoption.
The following 12 months will define Biosecure ID’s trajectory for the next decade.
If you believe that applied genomics and forensic science need better systems, not just better kits, this is the moment to act.
Join us — and help build what comes next.
We are raising £1m ($1.5m) in SEIS/EIS-eligible Seed funding, targeting close in March 2026, with a planned commercial launch in March 2027. This capital will accelerate the flywheel - turning momentum into global impact.
If you believe this market needs better systems — not incremental fixes — this is the moment to invest.
*Access to the Data Room is currently restricted to our present investors and with parties in advanced stages of investment discussions.
